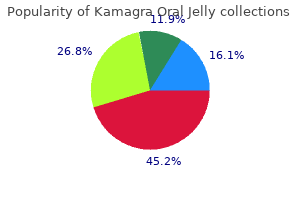
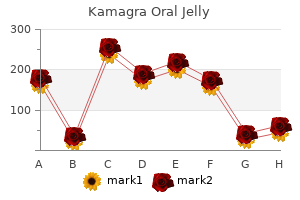
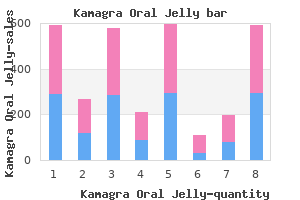
"Kamagra oral jelly 100 mg for sale, erectile dysfunction drugs bayer".
U. Ashton, M.S., Ph.D.
Co-Director, West Virginia School of Osteopathic Medicine
Experimental evidence In a yeast oestrogen screen assay erectile dysfunction treatment california buy discount kamagra oral jelly 100mg line, the fruit oil from aniseed was Aristolochia Aristolochia species (Aristolochiaceae) A Synonym(s) and related species the nomenclature of these and related plants has given rise to confusion with other thyroid causes erectile dysfunction 100 mg kamagra oral jelly for sale, non-toxic plants erectile dysfunction doctor pune kamagra oral jelly 100 mg free shipping. This has been exacerbated by the fact that different Chinese names have been used for each species impotence of organic nature purchase 100mg kamagra oral jelly with visa. Birthwort has been used as a collective name for the Aristolochia species, but it has also been used for one of the species, Aristolochia clematitis L. The Chinese name Mu Tong has been used to refer to some of the Aristolochia species. Aristolochia fangchi has been referred to by the Chinese names Fang Chi, Fang Ji, Guang Fang Ji. Constituents All species contain a range of toxic aristolochic acids and aristolactams. Use and indications Aristolochic acids and aristolactams are nephrotoxic, carcinogenic and cytotoxic. Numerous deaths have resulted from aristolochic acid nephropathy and associated urothelial cancer, caused by ingestion of aristolochia both medicinally and from contamination of food. All plants of the family Aristolochiaceae are banned in Europe and elsewhere, and should be avoided. Constituents the main constituents of the bark are triterpenoid saponins including arjunic acid, arjunolic acid, arjungenin and arjunglycosides, and high levels of flavonoids, such as arjunone, arjunolone, luteolin and quercetin. Polyphenols, particularly gallic acid, ellagic acid and oligomeric proanthocyanidins, are also present. Interactions overview Arjuna appears to have some effects on cardiovascular function, which may lead to interactions with conventional drugs used for similar indications. Arjuna may also affect thyroid function, which could alter the control of both hyperand hypothyroidism. For information on the interactions of individual flavonoids present in arjuna, see under flavonoids, page 186. Use and indications Arjuna is widely used in Ayurvedic medicine for the 36 Arjuna 37 Arjuna + Cardiovascular drugs Arjuna appears to have some effects on cardiovascular function that may be of benefit when given with conventional cardiovascular drugs. Clinical evidence the effect of arjuna on angina pectoris, congestive heart failure, left ventricular mass and hyperlipidaemia has been investigated in a number of small studies in patients with various cardiovascular disorders (these have been the subject of a review1). For example, in one double-blind, crossover study in 58 patients with stable angina, the addition of powdered stem bark extract (500 mg every 8 hours) for one week decreased the number of angina episodes and the need for nitrate therapy during episodes of angina (about 5. This improvement was maintained over long-term evaluation in an open phase, when patients continued the bark extract at the same dosage. Arjuna is purported to have inotropic and hypotensive effects, as well as lipid-lowering effects. See Arjuna + Thyroid and Antithyroid drugs below for the possibility that some of the cardiovascular effects of arjuna might occur via an antithyroid action. Importance and management Arjuna has been used in small numbers of patients taking a variety of conventional cardiovascular drugs, apparently without particular problems, and with possible additional benefit. Efficacy of Terminalia arjuna in chronic stable angina: a double-blind, placebo-controlled, crossover study comparing Terminalia arjuna with isosorbide mononitrate. Salutary effect of Terminalia Arjuna in patients with severe refractory heart failure. Antioxidant and hypocholesterolaemic effects of Terminalia arjuna tree-bark powder: a randomised placebo-controlled trial. Arjuna + Thyroid and Antithyroid drugs the interaction between arjuna and thyroid or antithyroid drugs is based on experimental evidence only. Experimental evidence In a study in animals, arjuna bark extract appeared to inhibit thyroid function. Giving levothyroxine increased the level of thyroid hormones, increased the heart to body weight ratio, as well as increasing cardiac and hepatic lipid peroxidation. When the plant extract was given simultaneously, the level of thyroid hormones, and also the cardiac lipid peroxidation, were decreased. These effects were comparable to those of a standard antithyroid drug, propylthiouracil.
Syndromes
- Post-pericardiotomy syndrome -- low fever and chest pain that can last for up to 6 months
- Etching cream
- Replacement of blood volume
- Heart stress test
- Lung cancer or cancer of another area of the body that has spread to the lungs
- Damage to, and failure of, many organ systems
- Wear trousers and long-sleeved shirts, particularly at dusk.
Pneumonia impotence bicycle seat buy cheap kamagra oral jelly 100 mg, colitis erectile dysfunction pump youtube buy kamagra oral jelly 100 mg fast delivery, retinitis erectile dysfunction doctors in atlanta 100mg kamagra oral jelly amex, and a syndrome characterized by fever impotence at 19 generic 100 mg kamagra oral jelly mastercard, thrombocytopenia, leukopenia, and mild hepatitis may occur in immunocompromised hosts, including people topoietic stem cell transplantation. Less commonly, patients treated with biologic response Congenital infection has a spectrum of clinical manifestations but usually is not evirestriction, jaundice, purpura, hepatosplenomegaly, microcephaly, intracerebral (typically is estimated to occur in 3% to 10% of infants with symptomatic infections, or 0. Between 55% and 75% of symptomatic and asymptomatic children, respectively, who versal newborn hearing screen will not detect the majority of infants who are at risk of ated regularly for early detection and appropriate intervention of suspected hearing losses. Infection acquired from maternal cervical secretions during the intrapartum period, or in the postpartum period from human milk, usually is not associated with clinical illness in term babies. Transmission occurs horizontally (by direct person-to-person contact with virus-containing secretions), vertically (from mother to infant before, during, or after birth), and via transfusions of blood, platelets, and white blood cells from infected a primary infection and intermittent virus shedding and symptomatic infection can occur throughout the lifetime of the infected person, particularly under conditions of immunosecretions from infected individuals, but contact with infected urine also can have a role. In addition, these children frequently including mothers who may be pregnant, and other caregivers, including child care staff hence, blood transfusions and organ transplantation can result in transmission. Severe recipients who receive transplants from seronegative donors are at greatest risk of disease pressed people and result in disease if immunosuppression is severe (eg, in patients with transplant recipients). Congenital infection and associated sequelae can occur irrespective of the trimester of pregnancy when the mother is infected, but severe sequelae are associated more Damaging fetal infections following nonprimary maternal infection have been reported, and acquisition of a different viral strain during pregnancy in women with preexisting are born to women with nonprimary infection, and the contribution of nonprimary remains contentious and is an active area of research. Similarly, although disease can occur in ingestion of infected human milk do not develop clinical illness or sequelae, most likely because of the presence of passively transferred maternal antibody. Among infants who acquire infection from maternal cervical secretions or human milk, preterm infants born the incubation period incubation period to the size of the virus inoculum and route of infection. It is important to note that standard virus cultures must be maintained for been associated with a fourfold antibody titer increase in paired serum specimens or by necessarily indicate acute infection or disease, especially in immunocompetent people. If an infant is unable to absorb medications reliably from the gastrointestinal tract (eg, because of necrotizing enterocolitis or other bowel disorders), intravenous with parenteral ganciclovir. Absolute neutrophil counts should be performed weekly for aminotransferase concentration should be measured monthly during treatment. If such patients are treated with parenteral ganciclovir, a clovir can be considered if symptoms and signs have not resolved. Foscarnet is more toxic (with high rates of limiting nephrotoxicity) ease caused by ganciclovir-resistant virus or people who are unable to tolerate ganciclovir. When caring for children, hand hygiene, particularly after changing be treated differently from other children. Pasteurization or freezing of donated human - antibody-negative women should be considered. For further information on human milk Prevention of Transmission in Transplant Recipients. Approximately 5% of patients develop severe dengue, which is more common with second or other subsequent infections. Less common clinical syndromes include myocarditis, pancreatitis, hepatitis, and neuroinvasive disease. Warning signs of progression to severe dengue occur in the late febrile phase and include persistent shock, and a rapid decline in platelet count with an increase in hematocrit. Patients with nonsevere disease begin to improve during the critical phase, but people with clinically disease with pleural effusions and/or ascites, hypovolemic shock, and hemorrhage. Because of the approximately 7 days of tissue; percutaneous exposure to blood; and exposure in utero or at parturition. Dengue is a major public health problem in the tropics and subtropics; an estimated 50 to 100 million dengue cases occur annually in more than 100 countries, and 40% cause of febrile illness among travelers returning from the Caribbean, Latin America, and A aegypti and 35 states have A albopictus mosquitoes, local dengue transmission is uncommon because of infrequent contact between people and infected mosquitoes. It is most likely to cause severe disease in young children and women, especially pregnant women, and in patients with chronic diseases (asthma, sickle cell anemia, and diabetes mellitus). In humans, the incubation period is 3 to 14 days before symptom onset (intrinsic incubation). Infected people, both symptoms develop and throughout the approximately 7-day viremic period. Additional supportive care is required if the patient becomes dehydrated or develops warning signs of severe disease at the time of fever defervescence. No chemoprophylaxis or antiviral medication is available to treat patients with dengue.
Cyclosporine A or intravenous cyclophosphamide for lupus nephritis: the Cyclofa-Lune study erectile dysfunction treatment in bangladesh generic 100mg kamagra oral jelly mastercard. Cyclophosphamide pharmacokinetics and dose requirements in patients with renal insufficiency impotence effects on marriage generic kamagra oral jelly 100 mg. Strategies for preservation of ovarian and testicular function after immunosuppression erectile dysfunction drugs singapore kamagra oral jelly 100 mg with mastercard. Use of a gonadotropinreleasing hormone analog for protection against premature ovarian failure during cyclophosphamide therapy in women with severe lupus impotence organic origin definition kamagra oral jelly 100mg with visa. Mycophenolate mofetil vs cyclophosphamide therapy for patients with diffuse proliferative lupus nephritis. Randomized controlled trial of pulse intravenous cyclophosphamide versus mycophenolate mofetil in the induction therapy of proliferative lupus nephritis. Is mycophenolate mofetil superior to pulse intravenous cyclophosphamide for induction therapy of proliferative lupus nephritis in Egyptian patients? Histopathologic and clinical outcome of rituximab treatment in patients with cyclophosphamideresistant proliferative lupus nephritis. Is combination rituximab with cyclophosphamide better than rituximab alone in the treatment of lupus nephritis? A retrospective seven-year analysis of the use of B cell depletion therapy in systemic lupus erythematosus at University College London Hospital: the first fifty patients. Rituximab in systemic lupus erythematosus: A systematic review of off-label use in 188 cases. Treating lupus: from serendipity to sense, the rise of the new biologicals and other emerging therapies. Predictive power of the second renal biopsy in lupus nephritis: significance of macrophages. Remission, relapse, and re-remission of proliferative lupus nephritis treated with cyclophosphamide. Lupus nephritis: prognostic factors and probability of maintaining lifesupporting renal function 10 years after the diagnosis. Prognosis in proliferative lupus nephritis: the role of socio-economic status and race/ethnicity. Predictors of relapse and end stage kidney disease in proliferative lupus nephritis: focus on children, adolescents, and young adults. Changes in antibodies to C1q predict renal relapses in systemic lupus erythematosus. Laboratory tests as predictors of disease exacerbations in systemic lupus erythematosus. Routine immunologic tests in systemic lupus erythematosus: is there a need for more studies? A decrease in complement is associated with increased renal and hematologic activity in patients with systemic lupus erythematosus. Effect of angiotensin-converting enzyme inhibitors on the progression of nondiabetic renal disease: a metaanalysis of randomized trials. Angiotensin-Converting-Enzyme Inhibition and Progressive Renal Disease Study Group. Very long-term outcome of pure lupus membranous nephropathy treated with glucocorticoid and azathioprine. Treatment of pure membranous lupus nephropathy with prednisone and azathioprine: an open-label trial. Randomized, controlled trial of prednisone, cyclophosphamide, and cyclosporine in lupus membranous nephropathy. Mycophenolate mofetil as the primary treatment of membranous lupus nephritis with and without concurrent proliferative disease: a retrospective study of 29 cases. Tacrolimus for the treatment of systemic lupus erythematosus with pure class V nephritis.
Facts on the ground erectile dysfunction treatment dallas purchase 100 mg kamagra oral jelly mastercard, together with advancements through principles of continuous improvement in relation to labour practices doctor for erectile dysfunction philippines kamagra oral jelly 100 mg with visa, at the various stages of the crop calendar impotence urologist generic kamagra oral jelly 100mg, continue to be clearly documented impotence natural remedies purchase 100mg kamagra oral jelly mastercard. Kindest Regards Marcus McKay Head of Corporate Responsibility & Communications T: +45 60 42 74 57 E: m. Swig Makoto Takano Marie Warburg Mike Roberts Director Curverid Tobacco P/L 91 Coventry Road Workington Harare Zimbabwe Re: Human Rights Watch research on tobacco farms in Zimbabwe Dear Mr. Roberts, We are writing to share preliminary findings from research that Human Rights Watch has carried out regarding human rights abuses on tobacco farms in Zimbabwe, and to seek your response. We interviewed more than 60 small-scale tobacco farmers, including some who said they produced tobacco leaf independently and sold it on auction floors, and some who produced and sold tobacco leaf through contracts with Curverid Tobacco or other tobacco companies. We documented hazardous child labor, as well as serious health and safety risks, labor rights abuses, failure to provide copies of contracts to contracted farmers, and other human rights problems, including on some farms supplying Curverid Tobacco. Human Rights Watch also interviewed more than a dozen children who worked for hire on tobacco farms of various sizes, as well as several young adults who started working in tobacco farming as children. Many workers reported that employers paid them with delays, from a few days up to weeks or months. Interviewees reported these symptoms while harvesting tobacco, performing tasks involved in the curing process, and sorting dried tobacco leaves. Farm Monitoring and Inspection Nearly all small-scale farmers who were producing and selling tobacco under contracts with tobacco companies, including Curverid Tobacco, reported that company representatives regularly visited their farms to share information and advice. Does Curverid Tobacco purchase tobacco from Zimbabwe, either directly or through subsidiaries or suppliers? Does Curverid Tobacco or its subsidiaries or suppliers contract directly with tobacco farmers or groups of farmers in Zimbabwe? If so, how many farmers were contracted with Curverid Tobacco in 2015, 2016, and 2017, and in which provinces? What proportion of the total tobacco purchased by Curverid Tobacco in 2015, 2016, and 2017 was purchased from contracted growers? Does Curverid Tobacco have a policy regarding the provision of copies of signed contracts to signatories? Does Curverid Tobacco or its subsidiaries or suppliers purchase tobacco from auction floors in Zimbabwe? What proportion of the total tobacco purchased by Curverid Tobacco in 2015, 2016, and 2017 was purchased through this system? How does Curverid Tobacco communicate its standards and expectations regarding child labor to growers and suppliers, including growers who may be selling tobacco leaf to Curverid Tobacco on auction floors? Has Curverid Tobacco identified or received any reports of child labor on tobacco farms supplying tobacco to Curverid Tobacco in Zimbabwe in 2015, 2016, or 2017? We would welcome any additional information Curverid Tobacco would like to provide to Human Rights Watch regarding its policies and practices toward eliminating child labor in tobacco farming in Zimbabwe. How does Curverid Tobacco communicate its standards and expectations regarding labor rights to growers and suppliers, including growers who may be selling tobacco leaf to Curverid Tobacco on auction floors? What steps does Curverid Tobacco take to protect tobacco farmers, their families, and hired workers in its supply chain from nicotine poisoning or Green Tobacco Sickness? How does Curverid Tobacco ensure workers in the supply chain are informed about the risks of nicotine poisoning or Green Tobacco Sickness? What policies does Curverid Tobacco have in place regarding handling and applying pesticides, disposal of pesticide containers, as well as the proximity of workers on tobacco farms in its supply chain to active spraying of pesticides or other hazardous chemicals? How does Curverid Tobacco ensure workers in the supply chain are informed about the risks of pesticide exposure? How does Curverid Tobacco monitor for child labor, labor rights abuses, health and safety violations, or other human rights problems in the supply chain? What due diligence policies and procedures does Curverid Tobacco have in place to identify, prevent, mitigate, and account for possible impacts of your company or your suppliers on human rights, including child labor and labor rights? How does Curverid Tobacco ensure that all your suppliers are using rigorous human rights due diligence measures? To that end, we would welcome a formal response to this letter by September 18, 2017.


